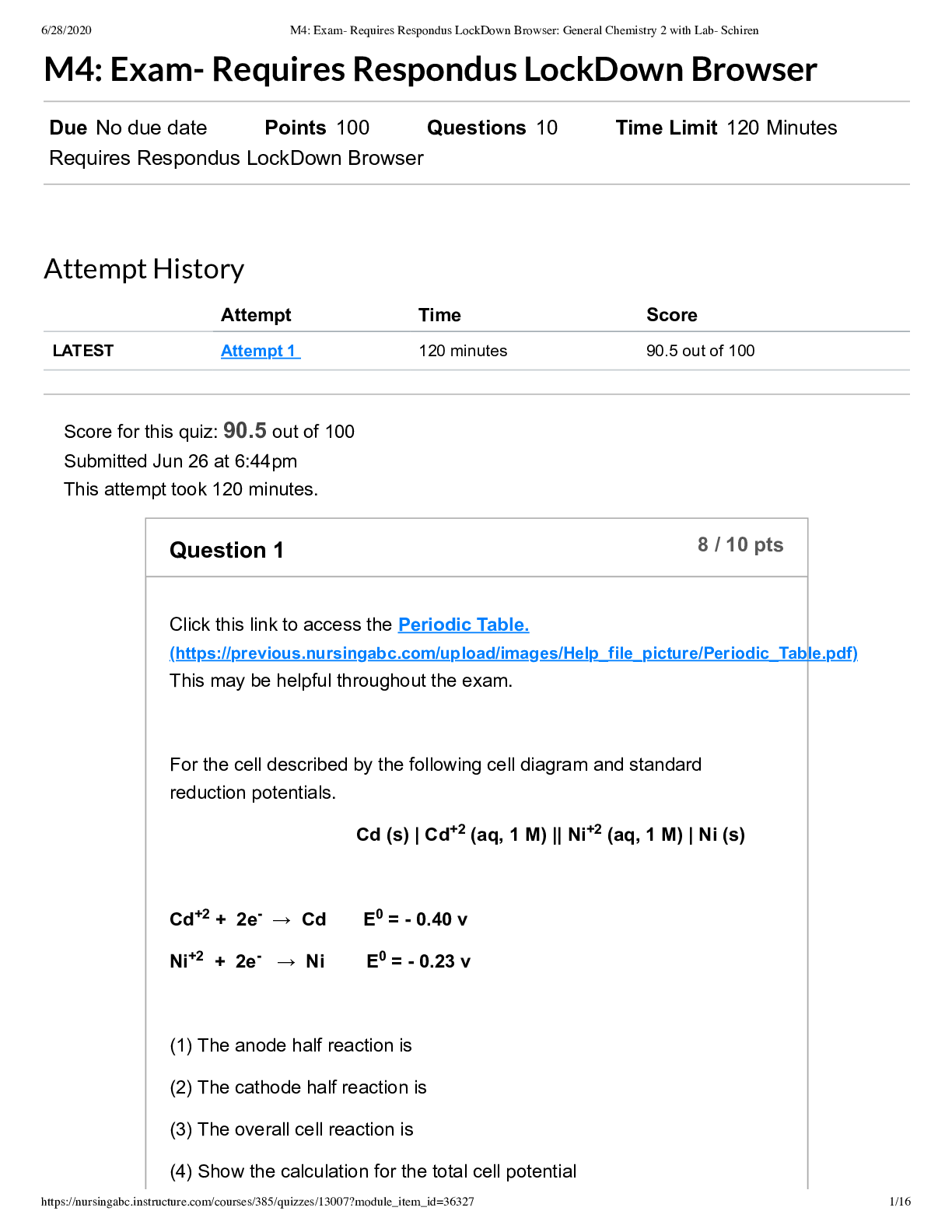
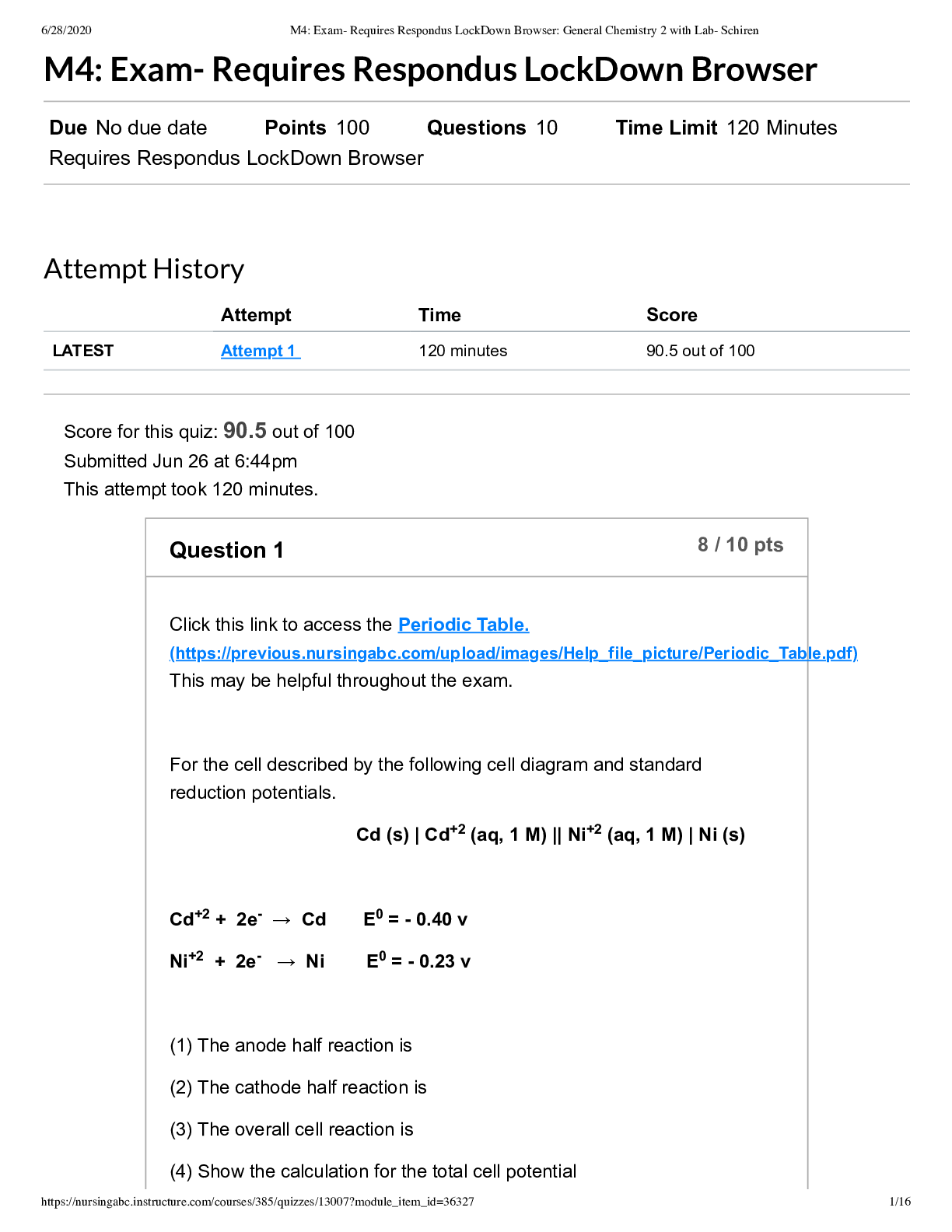
General Chemistry 2 with Lab- Schiren M4 Exam- Requires Respondus LockDown Browser
Course
English
Subject
Chemistry
Category
Questions and Answers
Pages
16
Uploaded By
ATIPROS
Preview 4 out of 16 Pages
Download all 16 pages for $ 8.50
Reviews (0)
$8.50
